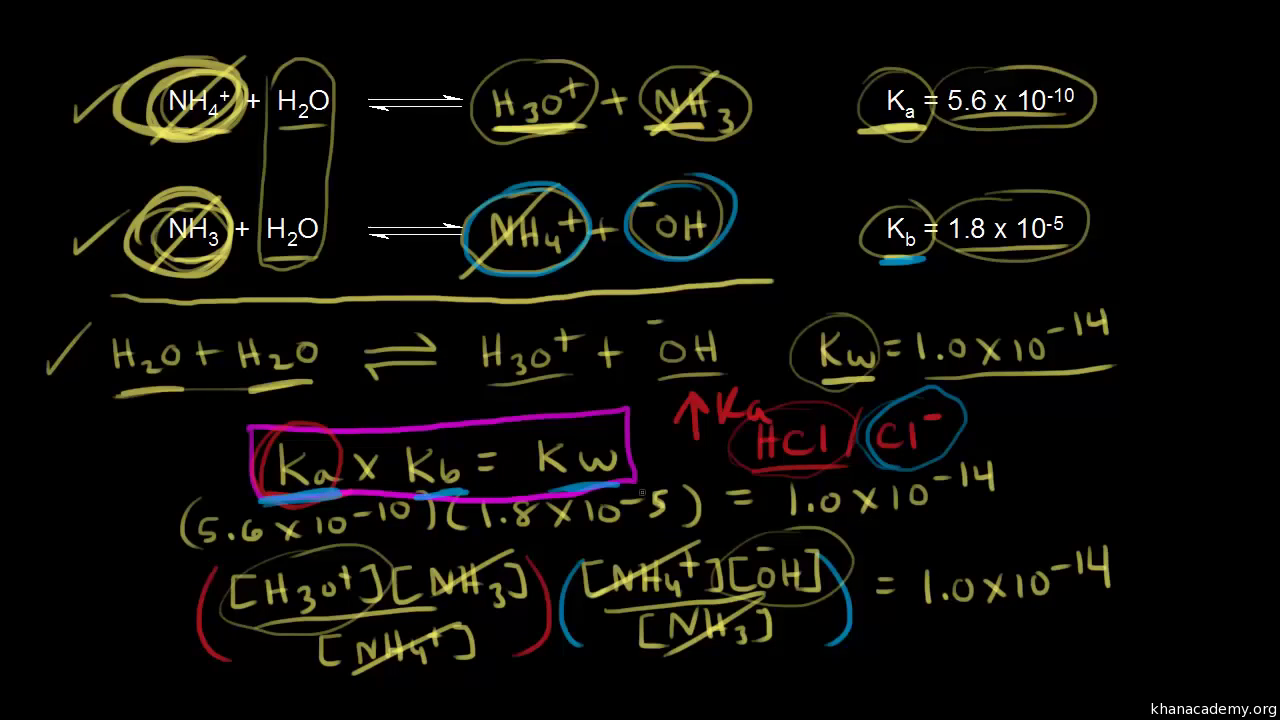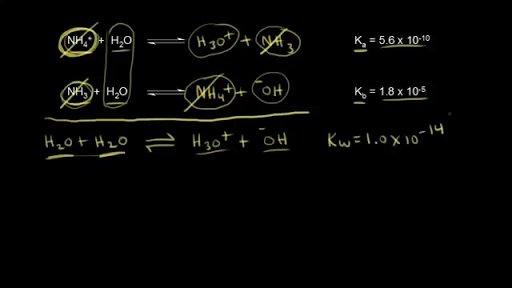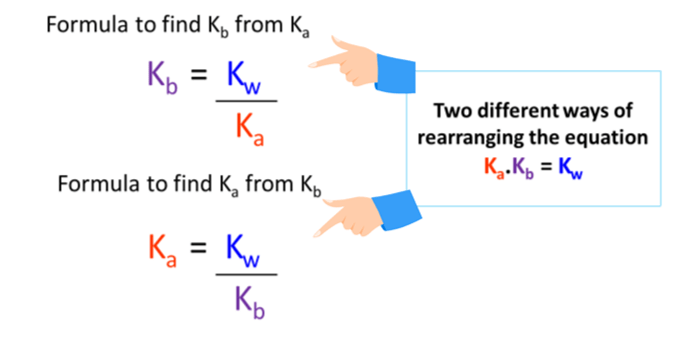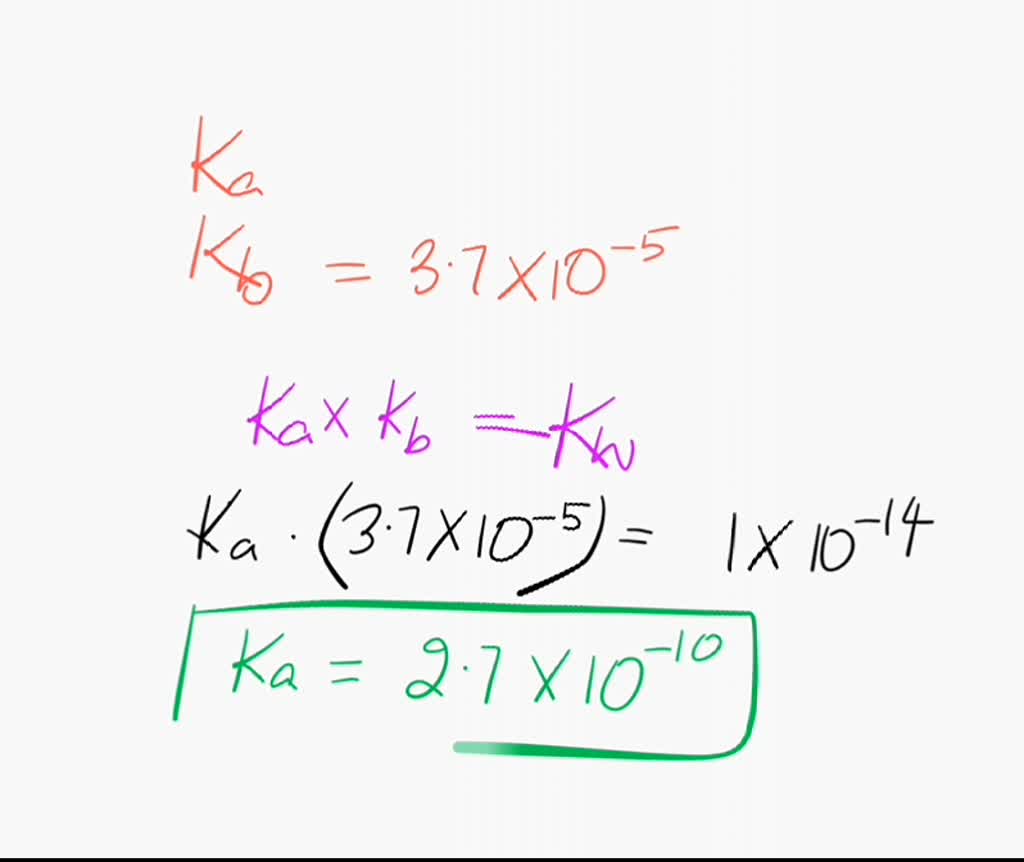
SOLVED: The Relation Between Ka and Kb of a Conjugate Acid-Base Pair A key relationship exists between the Ka of HA and the Kb of its conjugate base, A-, which we can

From the dissociation constants Ka and Kb for an acid and its conjugate base, show that Ka· Kb = Kw .
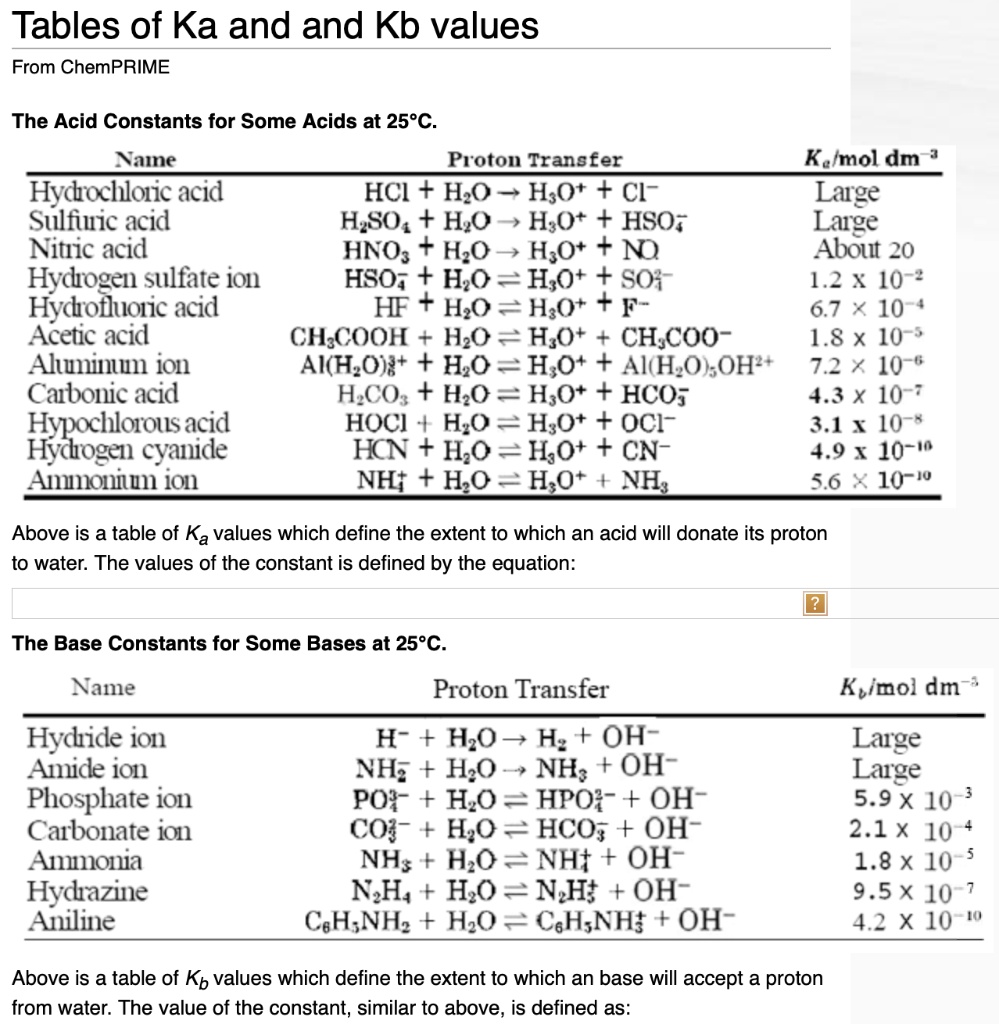
SOLVED: Tables of Ka and Kb values From ChemPRIME The Acid Constants for Some Acids at 25°C Name Proton Transfer Kalmol dm Hydrochloric acid HCl + H2O -> H3O+ + Cl- Large

How is Kw=Ka x Kb if each of those equilibrium constants include the concentration of another substance in them? Kw=H3o x oh but ka which is h3o also includes another substance? :